
Different possibilities were considered (see Supplementary Notes), but we found enzymatically catalysed gas formation from dissolved precursors most promising. 1.1 g mL −1 for an nLR containing mostly water and cells 10 to less than 0.9 g mL −1. We reasoned that in order to readily separate an nLR containing a microcolony of a desired strain variant from a background of nLRs, we should aim for a difference in buoyancy of at least 20%, i.e. 10 to 100 µm colony diameter), analysis, and recovery of positive variants 6, 7, 8, 9. As cultivation vessels, we used monodisperse hydrogel-based nanolitre reactors (nLRs), with a precisely adjustable diameter between 200 to 500 µm, as microcompartments for controlled expansion of single cells into monoclonal microcolonies (approx. By changing the buoyancy force of a cultivation vessel with a monoclonal microcolony in response to a desired change, such vessels can be made to ascend from a background population and float, allowing for strictly parallel analysis and sorting of strain variants at very high rates. We exploited Archimedes’ principle, which relates the buoyancy force of an immersed object to its density, to identify positive variants in biocatalyst screening and selection assays. Methods enabling high throughput at reasonable costs are therefore in high demand 2, 3, 4, 5. As a result, the throughput remains limited. 
Frequently, analysis and sorting of the samples occur in a consecutive fashion in which samples are analysed one after the other, and then a mechanical operation is applied to separate a potentially positive variant from the rest. For screening assays, improved strains are typically identified by a colour change, or an increased fluorescence or luminescence signal, while in the case of selection assays, positive variants show an elevation of the growth rate over the background of negative variants 1. In both cases, changes in the concentration or the activity of the product need to be read out. Screening and selection procedures are key strategies in the discovery or optimization of whole cell biocatalysts in high-throughput campaigns. We demonstrate the potential of the method for both, screening and selection protocols, and envision a wide applicability of the system for biosensor-based assays. 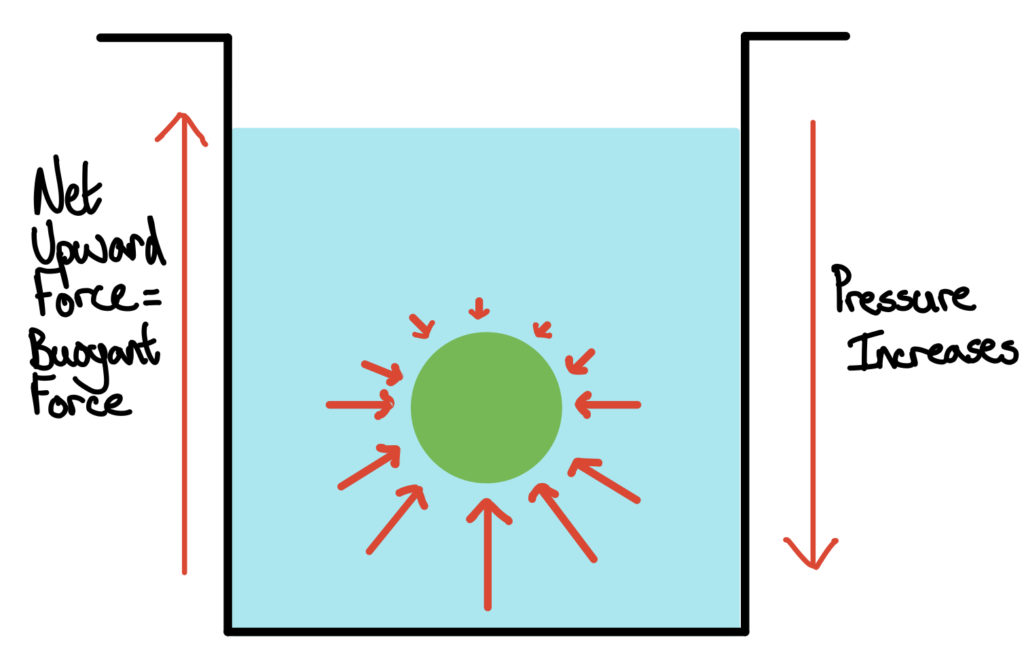
Exploiting Archimedes’ principle, this density shift and the resulting upward buoyancy force can be used for batch-wise library sampling. Exposure to hydrogen peroxide leads to oxygen gas formation and thus to a density shift of the cultivation vessel. Specifically, product formation in nanolitre-sized cultivation vessels is sensed and translated into the formation of catalase as a reporter protein. We present a method for space- and time-efficient processing of very large libraries (10 7) of recombinant cellular catalysts, in which the phenotypic characterisation and the isolation of positive variants for the entire library is done within one minute in a single, highly parallelized operation. Frequently, large libraries of semi-rationally generated variants are sampled in high-throughput mode in order to then identify improved catalysts. The complex reaction network employed by cellular catalysts and the still only moderate predictive power of modelling approaches leaves this tool challenging to engineer. The ability of whole cells to catalyse multistep reactions, often yielding synthetically demanding compounds later used by industrial biotech or pharma, makes them an indispensable tool of synthetic chemistry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
